 
Note that each of these entries corresponds to one of the twelve periods respectively. Group six (R H superscript two, R 0 superscript three) contains the following information: (no entry for period 1), O = 16, S = 32, C r = 52, S o = 78, M o = 96, T o = 125, -, -, W = 184, -, U = 240. Group five (R H superscript two, R superscript two 0 superscript five) contains the following information: (no entry for period 1), N = 14, P = 31, V = 51, A s = 75, N b = 94, S b = 122, -, -, T a = 182, B l = 208,. Group four (RH superscript four, R0 superscript eight) contains the following information: (no entry for period 1), C = 12, B i = 28, T i = 48, - = 72, Z r = 90, S n = 118, ? C o = 140, ? L a = 180, P b = 207, T h = 231. = 44, - = 68, ? Y t = 88, I n = 113, ? D I = 138, -, ? E r = 178, T l = 204, -. Periodic Table of Elements (Interactive) PDF - This Interactive Periodic Table is a tabular arrangement of the chemical elements, organized based on chemical element names, symbols, atomic masses, electron configuration and other chemical properties. 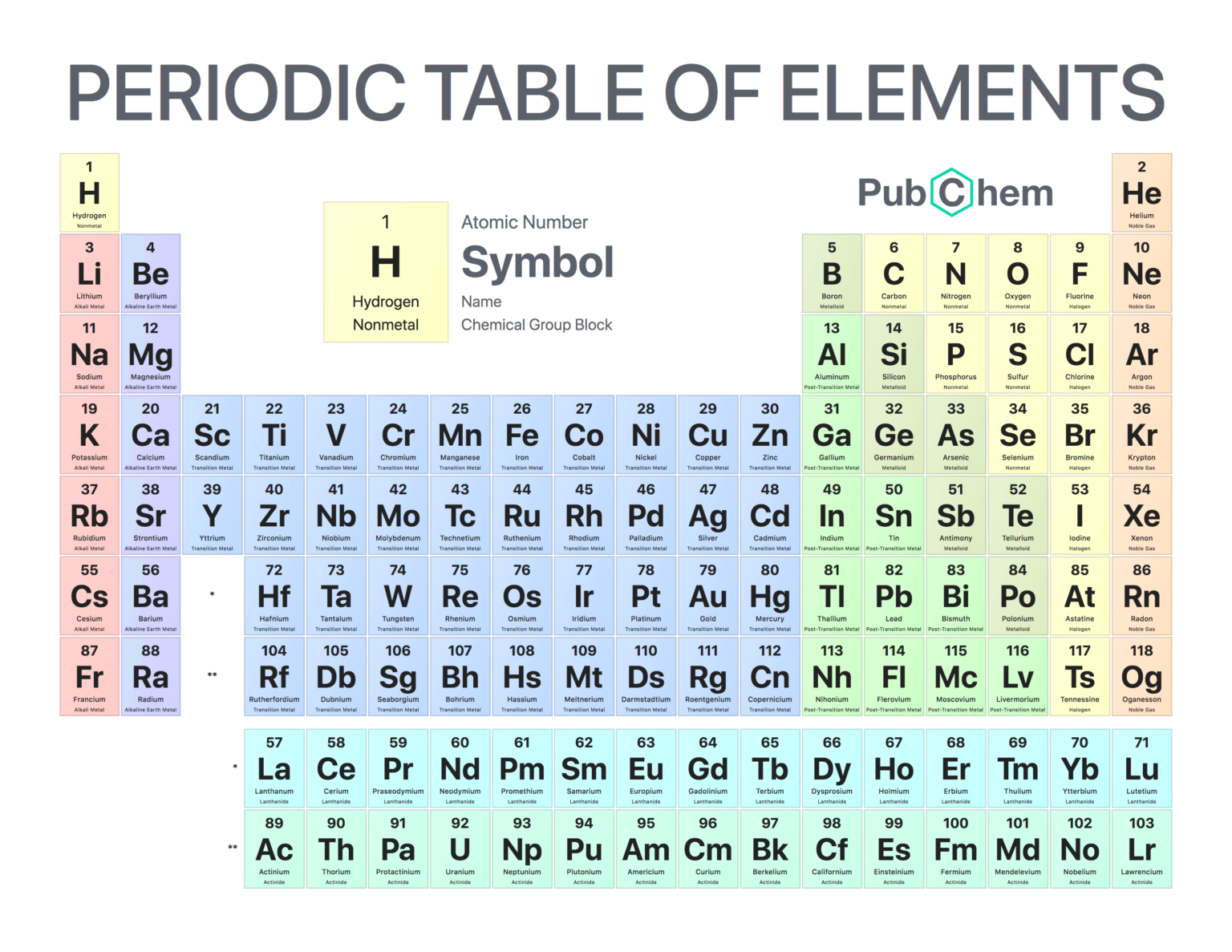
Group three (-, R superscript one 0 superscript nine) contains the information: (no entry for period 1), B = 11, A l = 27, 8. Period A horizontal row in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. Group A vertical column in the periodic table. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Members of a group typically have similar properties and electron configurations in their outer shell. Origin of the name: The name is derived from the Latin cadmia, the name for the mineral calamine. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. Note the ach of these entries corresponds to one of the twelve periods respectively. Origin of the name: The name is derived form the Latin fluere, meaning to flow Allotropes: F 2 F Fluorine 9. The second group (-, R 0) contains the following information: (not entry for period 1) B o = 9, 4, M g = 24, C a = 40, Z n = 65, S r = 87, C d = 112, B a = 187, -, -, H g = 200,. Figure B shows the first periodic table developed by Mendeleev, which had eight groups and twelve periods. Interactive Periodic Table of Elements Fisher Scientific Hydrogen. (credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons) Figure A shows a photograph of Dimitri Mendeleev. \): (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
